Scientists studying sulfate-rich terrains on Mars believe they may have identified a previously unknown mineral hidden in layered deposits near the planet’s vast canyon system. By combining spacecraft observations with laboratory experiments, researchers traced unusual spectral signals to a rare iron sulfate compound.
Sulfate minerals are common on Mars and often form when water evaporates, leaving behind chemical residues in the soil and rock. Because the planet’s surface is extremely dry today, many of these deposits have remained preserved for billions of years, allowing scientists to reconstruct past environmental conditions from orbit.
For years, planetary scientists analyzing data from NASA spacecraft noticed unusual spectral signatures in layered iron sulfates near Valles Marineris, one of the largest canyon systems in the solar system. These signals puzzled researchers for nearly two decades, as they did not correspond to any known mineral structure.
Unusual Signals on Mars
The investigation was led by Dr. Janice Bishop, a senior research scientist at the SETI Institute and NASA’s Ames Research Center. Her team combined orbital observations with laboratory experiments to understand the strange spectral patterns recorded by the CRISM instrument aboard NASA’s Mars Reconnaissance Orbiter.
According to Nature Communications, the data pointed to a rare compound called ferric hydroxysulfate, an iron sulfate phase that may represent a mineral not previously documented in nature. Scientists detected the compound by comparing its infrared absorption properties in laboratory experiments with the spectral signatures observed from Mars.
Dr. Bishop explained that the research focused on areas where layered sulfates and unusual spectral bands appeared together, suggesting that specific chemical processes had altered the minerals after their original formation.
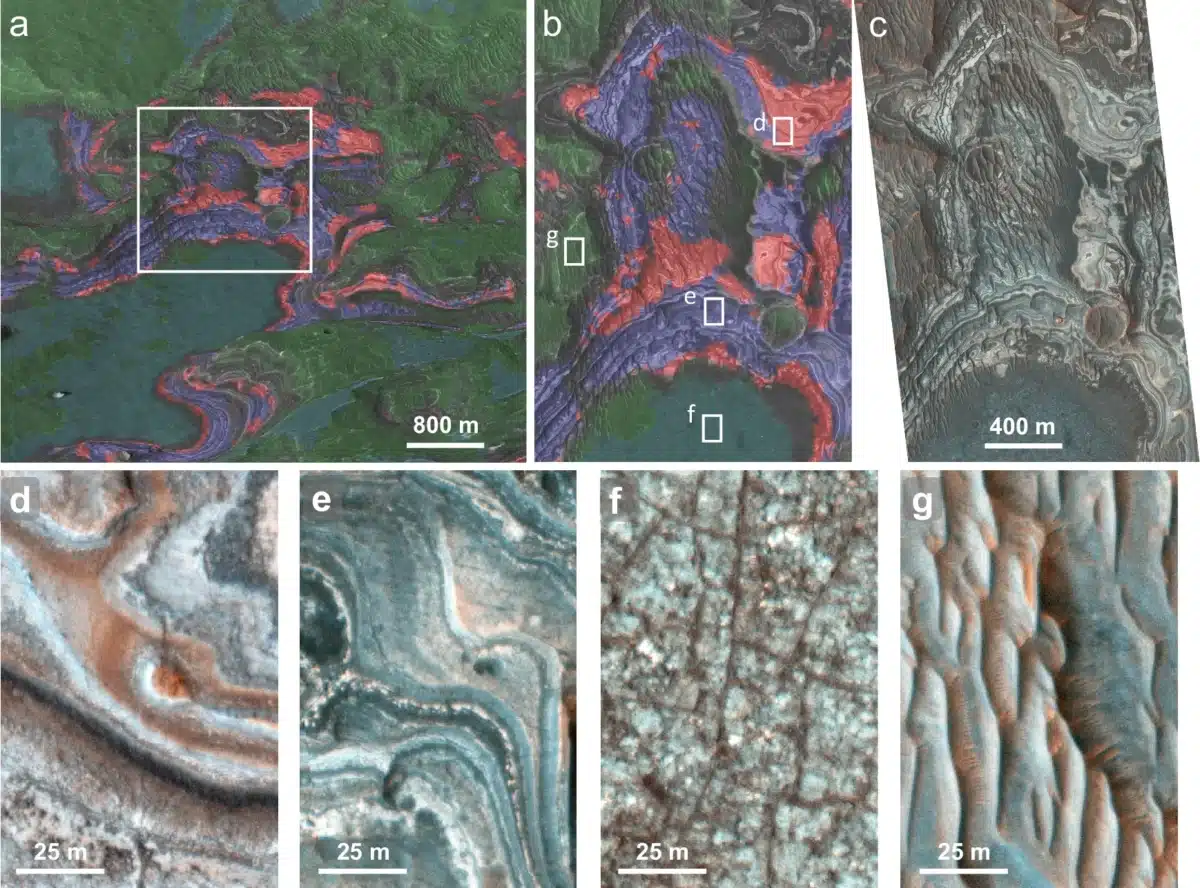
Two Regions Reveal Traces Of Ancient Water
Researchers concentrated on two locations near Valles Marineris: Aram Chaos, situated northeast of the canyon system, and the Juventae Plateau above Juventae Chasma, a canyon about five kilometers deep.
Both areas contain geological features associated with past water activity. As explained by the researchers, ancient channels cut across the Juventae Plateau, indicating that water once flowed through the region before evaporating and leaving sulfate-rich deposits behind.
These deposits occur in thin layers roughly one meter thick and lie between basaltic rock units. Planetary scientist Dr. Catherine Weitz noted that;
“Investigation of the morphologies and stratigraphies of these four compositional units allowed us to determine the age and formation relationships among the different units.”
Heat-driven Chemistry On Mars
Laboratory experiments helped researchers understand how the unusual mineral could have formed. The transformation begins with rozenite, an iron sulfate containing four water molecules in its structure. When heated to about 50°C, it converts into szomolnokite, which contains only one water molecule.
When temperatures rise above 100°C and the mineral is exposed to oxygen, the reaction produces ferric hydroxysulfate. This process alters the mineral’s atomic structure and its infrared signature, allowing scientists to identify it using orbital instruments.
The study also indicates that Mars still contains enough oxygen in its thin atmosphere for this reaction to occur. As explained by Dr. Johannes Meusburger, a postdoctoral researcher at NASA Ames:
“Our experiments suggest that this ferric hydroxysulfate only forms when hydrated ferrous sulfates are heated in the presence of oxygen,” he said. “While the changes in the atomic structure are very small, this reaction drastically alters the way these minerals absorb infrared light, which allowed identification of this new mineral on Mars using CRISM.”
The presence of ferric hydroxysulfate suggests that geothermal or volcanic heat may have altered sulfate deposits long after they originally formed, leaving behind a mineral phase that scientists may have detected for the first time on the Martian surface.
First Appeared on
Source link







Leave feedback about this