The outer curvature endures great strain: Located directly opposite the valve through which blood enters the heart, it gets hit with incoming blood. Researchers presumed that certain genes were more active in this part of the heart, and that this activity explained the formation of the muscular network of trabeculae. But when Priya and her student Christopher Chan looked, “none of the enzymes were there in the right time [or the] right place,” Priya said. Their absence left the researchers with one alternative to genetic instructions: physics.
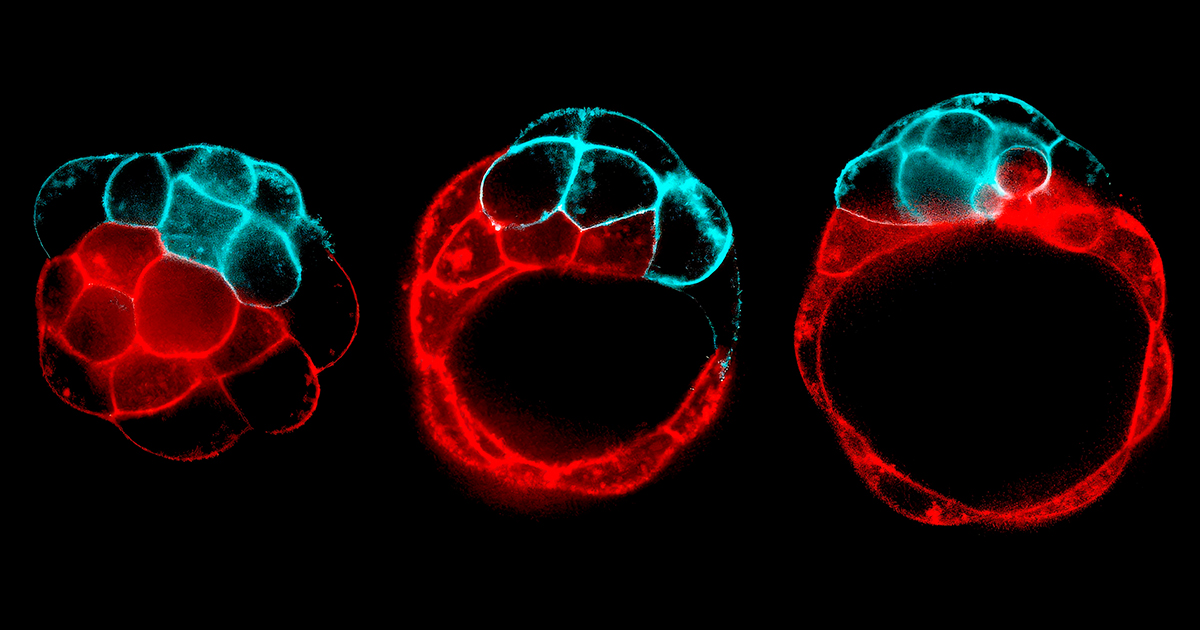
Priya and Chan imaged zebra fish hearts as they formed, capturing up to 100 frames per second at various intervals to closely follow any changes in their shape. Just six hours after the heart started beating, they noticed prominent gaps in the outer curvature’s cardiac jelly, a solid network of proteins that props up heart tissue. These gaps spread like fractures, and one day after they appeared, trabeculae strands started to knit their way across them. That timing suggested that these gaps might have something to do with how trabeculae form.
They asked the computational scientist Daniel Santos-Oliván on Torres-Sánchez’s team to develop simulations of beating hearts, which revealed that the gaps in the cardiac jelly were indeed fractures. The model showed that as the heart pulses and takes shape, strain concentrates in the outer curvature, stretching and contracting the jelly scaffold so much that it thins, weakens, and eventually breaks. Sensing those fractures, heart muscle cells at the outer curvature then peel away from the heart wall and fall into the jelly’s newly formed cracks, where they seed the trabeculae. Without the simulations, “we could never have thought that [this process] is controlled by geometry,” Priya said.
Studies by Christopher Chan (left) and Rashmi Priya (right) showed how controlled fracture helps to build a heart that can withstand forces.
To test that hypothesis, the researchers sped up the zebra fishes’ heart rate; more fractures formed in the cardiac jelly. When they slowed the hearts, fewer cracks appeared. This confirmed that the fracturing was a physical process dependent on the incredible strain of heart contractions. To test it another way, the team engineered hearts to grow straight and tubular; when they did so, the fractures changed their direction, too. That behavior is “characteristic of a fracture,” Torres-Sánchez said, and “made us sure that these were [fractures] we were looking at.”
Priya’s team has since seen these fractures in the hearts of chicken embryos. Based on these preliminary findings, “it is tempting to speculate that human hearts might be shaped by similar structural processes,” she said. In any case, their zebra fish work, which is undergoing peer review, shows that a fundamental feature of an essential organ is shaped by mechanical forces ahead of genetics.
The recent revelation that fracturing can be a constructive tool during development doesn’t mean the phenomenon is particularly common. Still, it is widespread in animals: Ruptures, tears, and cracks are known to shape zebra fish nostrils, hydra mouths, fruit fly legs, and whole flatworms. And yet because fracturing seems so destructive, almost antithetical to growth, it has only recently been recognized as a way of shaping tissues, summed up in the recent review paper co-authored by Priya, Torres-Sánchez, Santos-Oliván, and Chan.
Maître suspects that more examples of fracturing will be found now that researchers know to look for it. In recent years, researchers have discovered that, aside from fracturing, living tissues also crumple, buckle, wrinkle, and fold via physical mechanisms that produce incredible diversity with just a few slight tweaks in tissue properties and architecture. “This is really far-reaching,” Milinkovitch said. Mechanics makes evolution “much easier to understand.”
First Appeared on
Source link





Leave feedback about this