Scientists have built an artificial neuron that operates at the same voltage range as living nerve cells and can respond to signals produced by real tissue.
That achievement closes a long-standing gap between electronic circuits and biological systems, allowing devices to communicate with living cells using the same electrical language.
Artificial neurons in the lab
Inside a laboratory circuit, the artificial neuron produced electrical spikes near 0.1 volts, closely matching the signals used by natural neurons.
By observing those signals, Jun Yao and colleagues at the University of Massachusetts Amherst (UMass Amherst) demonstrated that the device reproduced the voltage levels, timing patterns, and energy use typical of biological neurons.
Earlier artificial neurons could imitate some neural behavior but relied on much stronger electrical signals that prevented direct interaction with living cells.
Operating within the same electrical limits as biological neurons removes that barrier and opens the possibility of circuits that can link directly with cellular activity.
Why voltage mattered
Living neurons usually fire between roughly 70 and 130 millivolts, while many artificial versions have needed 0.5 volts or more.
“Previous versions of artificial neurons used 10 times more voltage, and 100 times more power, than the one we have created,” said Yao.
That gap wastes energy and makes direct contact harder, because stronger signals can overwhelm delicate cellular activity instead of matching it.
By bringing the voltage down, the new neuron cleared the main barrier that had kept bio-inspired hardware mostly theoretical.
Bacteria build the core
At the center sat a memristor, a tiny component whose resistance changes with current, tuned by bacterial protein nanowires to operate at biological voltage levels.
Those nanowires came from Geobacter sulfurreducens, a microbe already known for moving electrons outside its cells.
In repeated tests, the switch flipped on near 60 millivolts and 1.7 nanoamps, then reset instead of staying locked.
That self-resetting behavior gave the team a way to imitate the rise and fall of a real neural spike.
Circuits learned restraint
Once the memristor turned on, a capacitor charged quickly and pushed the output into a brief voltage spike.
As charge built up, the circuit forced the switch back off and created a refractory period, a short reset before another spike.
Because of that pause, each burst returned near zero instead of smearing into one long signal.
The output could then trigger another artificial neuron, which moves the design closer to usable networks than earlier one-shot devices.
Chemistry changed firing
Chemical cues also altered the device through neuromodulation, chemical tuning that changes how easily cells fire.
When sodium levels rose, one sensor sped the circuit’s reset step, and the artificial neuron fired more often.
With dopamine, a graphene sensor produced a two-way response, increasing activity at some doses and easing it at others.
That mattered because living brains depend on chemical context, not just raw electrical pulses, to route information.
Living cells and artificial neurons
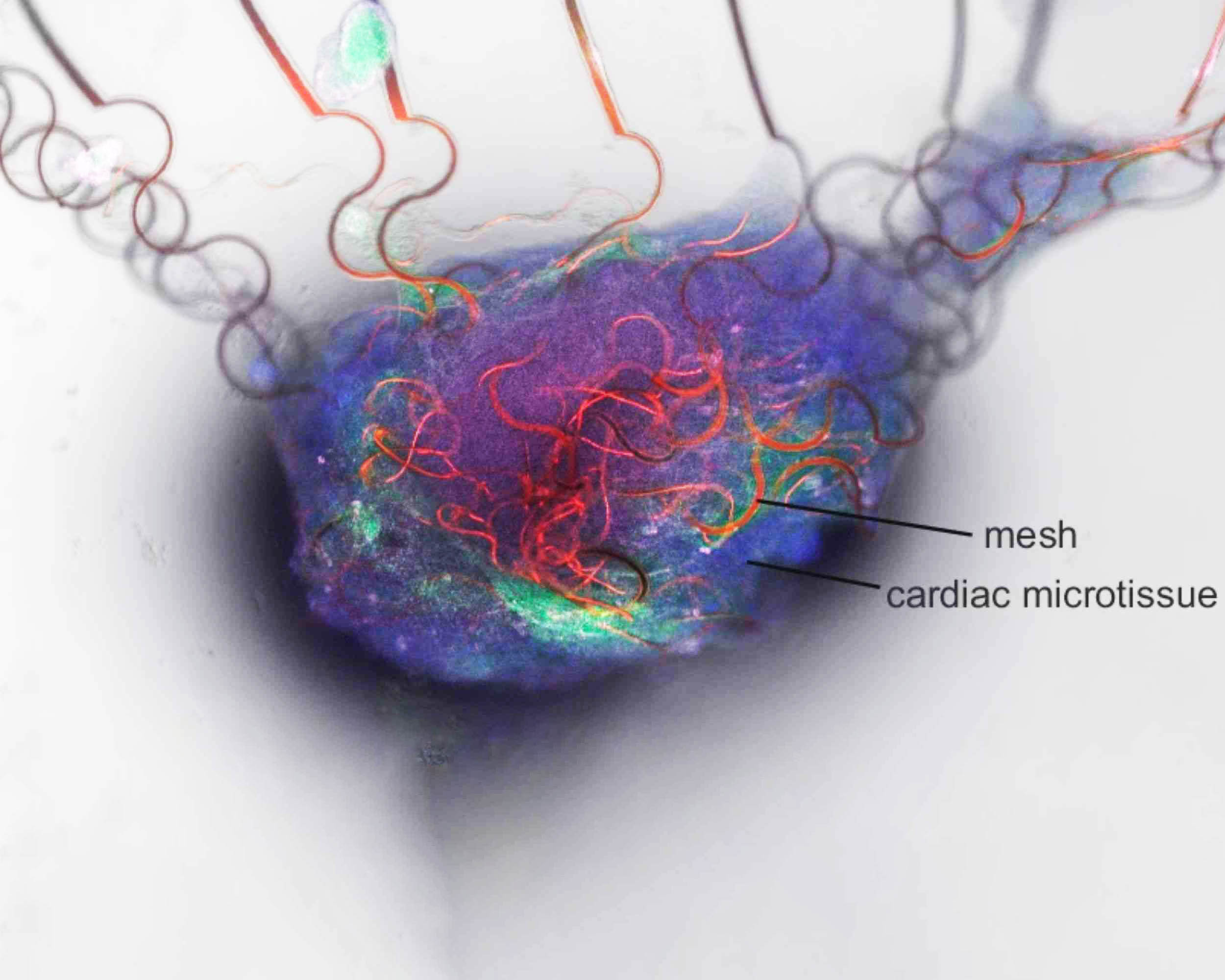
To test a living partner, the team linked the circuit to cardiomyocytes, heart muscle cells that beat through electrical signals.
Growing tissue wrapped around a soft mesh of graphene sensors, which picked up each cell’s electrical firing and contraction.
Normal activity in the heart cells left the artificial neuron silent, but a drug that sped up their rhythm triggered electrical spikes in the circuit.
That result did not prove a human brain link, but it did prove real-time conversation with living cells.
Wearables lose the middleman
Today’s wearable sensors often boost faint body signals before software can read them, which costs energy and adds hardware.
“That intermediate step of amplification increases both power consumption and the circuit’s complexity, but sensors built with our low-voltage neurons could do without any amplification at all,” said Yao.
The paper backs that point by showing the neuron can process real signals captured directly from neurons.
If sensing hardware improves enough, future patches or implants could become smaller, cooler, and easier to power.
Later versions stripped away extra sensing parts and still kept the same spiking energy, which made the design less bulky.
In that simpler form, chemical operation used at least 100 times less energy than earlier chemical artificial neurons.
Standard silicon-friendly fabrication should also help, because the whole circuit can be built with processes chip makers already use.
Even so, a prototype on a bench is not yet a medical device or a finished computer architecture.
A different computing path
What makes this work stand out is not one flashy trick, but the tight match between electronic and biological behavior.
Earlier devices often copied only the rough shape of a spike, while this one also matched voltage, energy, timing, and chemistry.
That broader match gives engineers a cleaner base for machines that sense, decide, and react near the body.
Much more testing still lies ahead, especially with true neurons and long-term stability, before anyone can promise implants or brain links.
Next steps for artificial neurons
By speaking in signals that living cells already use, the artificial neuron made efficiency and compatibility part of the same story.
Future progress will depend on better sensors, longer tests, and proof in nerve tissue, but the boundary looks less rigid now.
The study is published in Nature Communications.
—–
Like what you read? Subscribe to our newsletter for engaging articles, exclusive content, and the latest updates.
Check us out on EarthSnap, a free app brought to you by Eric Ralls and Earth.com.
—–
First Appeared on
Source link






Leave feedback about this